Nanostructure and cleaning mechanism of innovative media for polymer removal from artworks: a SANS study.
By Piero Baglioni
Remediation tools for conservation
In the past, synthetic polymers have been improperly applied as protective coatings to painted surfaces. Instead of preserving the paintings, these substances promoted a series of complex degradation mechanisms, which can ultimately lead to the destruction of the artwork, as shown in figure 1. Therefore the removal of these polymer films is one of the top priorities in Conservation Science.
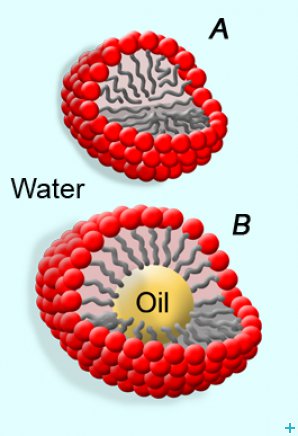
Micelles (See figure 2.A) and microemulsions (See figure 2.B) are among the most effective systems available for the optimal intervention in these cases, and represent an advanced methodology for polymer removal from porous materials such as mural supports, largely preferable to the use of organic solvent mixtures. Aqueous-based systems avoid the re-deposition of polymers inside the porous matrix of the artifact, and also reduce the toxicity of the application, offering at the same time better and more controlled results than neat organic solvents.
Nanostructure of cleaning systems
To design efficient nanofluids, prior knowledge of their ability to remove given polymer films is required. It is therefore essential to determine both their structure and dynamics. Complex nanofluids can be characterised by Small Angle Neutron Scattering (SANS). SANS is particularly suited for multi-component systems where different regions of the nanofluid can be labeled by isotopic substitution. Using Contrast variation, it is possible to characterise different components and/or regions of the aggregates, without changing the chemical composition and properties of the system.
Here we report on the structural characterisation of two nanofluids (EAPC and XYL [1-5]), which are both nanostructured systems based on sodium dodecylsulphate (SDS)/1-pentanol (PeOH). EAPC also contains Ethyl acetate (EA) and propylene carbonate (PC), both partially water-soluble solvents (EA ~8.3 % w/w; PC ~20 % w/w), while the XYL system contains p-xylene that is almost immiscible with water. Knowing the different properties of these compounds is essential to define the nano-structure of the aggregates. The interaction of these two nanofluids with Paraloid B72 (ethyl methacrylate/methyl acrylate 70:30) was studied through the structural determination of both nanofluids before, during and after the cleaning process.
Small-angle neutron scattering measurements were performed on the PAXE instrument of the Laboratoire Léon Brillouin, LLB, (Saclay, Gif-Sur-Yvette, France). Micelles were modeled as charged prolate ellipsoidal particles interacting with each other according to a screened Coulomb potential described by the classic NAR-MMSA model.
Structure and kinetics for effectiveness
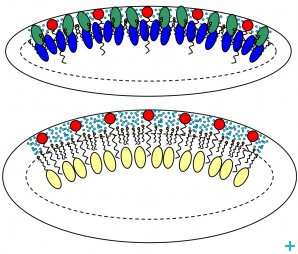
We found that the EAPC system contains elongated aggregates, dispersed in a mixture of water, PC, EA and PeOH, which cannot be considered simple ‘microemulsion droplets’, since both PeOH and PC have a co-surfactant action, while ethyl acetate is interposed between apolar tails and in the core of SDS. On the other hand, the XYL system is a classically defined microemulsion composed of a surfactant, a co-surfactant, and oil (i.e. xylene) contained in the core of the aggregate and separated from the aqueous continuous phase by a surfactant/co-surfactant layer.
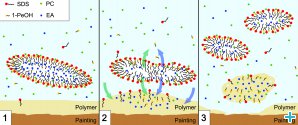
We performed many experiments and measurements to study the interaction mechanisms of EAPC and XYL with Paraloid B72 and get a complete, satisfactory picture of the phenomenon. We observed that the removal process is the same for both systems at the macro-scale, while there are significant differences in the kinetics of the process itself. In particular, EAPC is sensibly faster than XYL in interacting with polymer coatings. This behaviour can be explained by focusing on the interaction mechanism of the two systems. According to the results obtained from SANS we suggest that EAPC interacts with the polymer according to this model (see also figure 3):
1. Solvents dissolved in the continuous aqueous phase quickly interact with the polymer coating.
2. A series of exchanges occurs, where solvents migrate from the aqueous phase to the polymer, from the nanodroplets to the aqueous phase and from the nanodroplets to the polymer.
3. The polymer detaches from the substrate, which now is clean, while the nanodroplets get smaller and re-organize their structure due to the outflow of the solvents.
We proposed a similar mechanism for the XYL system, with one main difference, as the first step of the process is missing. In this process, solvents only migrate from the nanodroplets to the polymer to reach the equilibrium. The polymer then detaches from the substrate, while the nanodroplets get smaller and regain their structure.
Therefore, we conclude that the final step of both mechanisms is identical. The differences in the first steps, which are due to the different composition and consequent nano-structure of the systems, account for the different reaction speed that we observed. The speed of reaction indeed plays an important role in the effectiveness of these nanofluids in polymer removal from wall paintings, during real applications. We observed that, for an equal duration of application, EAPC is more effective than XYL in removing acrylic coatings.
We believe that a deeper understanding of the nano-structure and the mechanism that lies behind the cleaning process is the key of a more conscious approach to new conservation challenges.
This work was carried out together with Michele Baglioni and Debora Berti from the University of Florence, Italy.
References
[1] R. Giorgi, M. Baglioni, D. Berti, P. Baglioni, Accounts of Chemical Research, 2010, 43, 695–704.
[2] M. Baglioni, D. Rengstl, D. Berti, M. Bonini, R. Giorgi, P. Baglioni, Nanoscale, 2010, 2, 9, 1723-1732.
[3] M. Baglioni, R. Giorgi, D. Berti, P. Baglioni, Nanoscale, DOI: 10.1039/c1nr10911a.
[4] E. Carretti, L. Dei, P. Baglioni, Langmuir, 2003, 19, 7867-7872.
[5] M. Baglioni, D. Berti, J. Teixeira, R. Giorgi, P. Baglioni, submitted to Langmuir.
Piero Baglioni is a Full Professor of Physical Chemistry at the Department of Chemistry and CSGI of the University of Florence.